Hydromorphone and insulin cited twice as often as any other medication in harm incidents
A system developed to inform and support actions to improve medication safety for Canadians performed an analysis of medication incidents over a five-year span from 2015 to 2020. The analysis appeared in a recent Institute for Safe Medication Practices Canada (ISMP Canada) bulletin (Volume 20 • Issue 11).
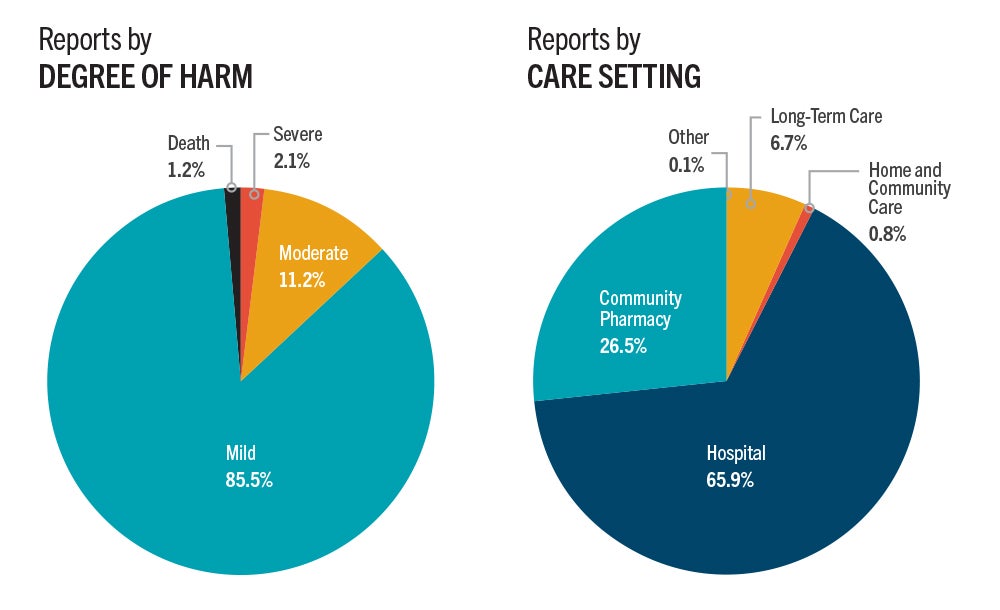
The analysis included a total of 7,531 harm incidents from databases operated by ISMP Canada and CIHI. A key finding is nearly 86 percent of all harm incidents were reported as mild in severity. Reports from hospitals form most of the incidents, followed by those from community pharmacies.
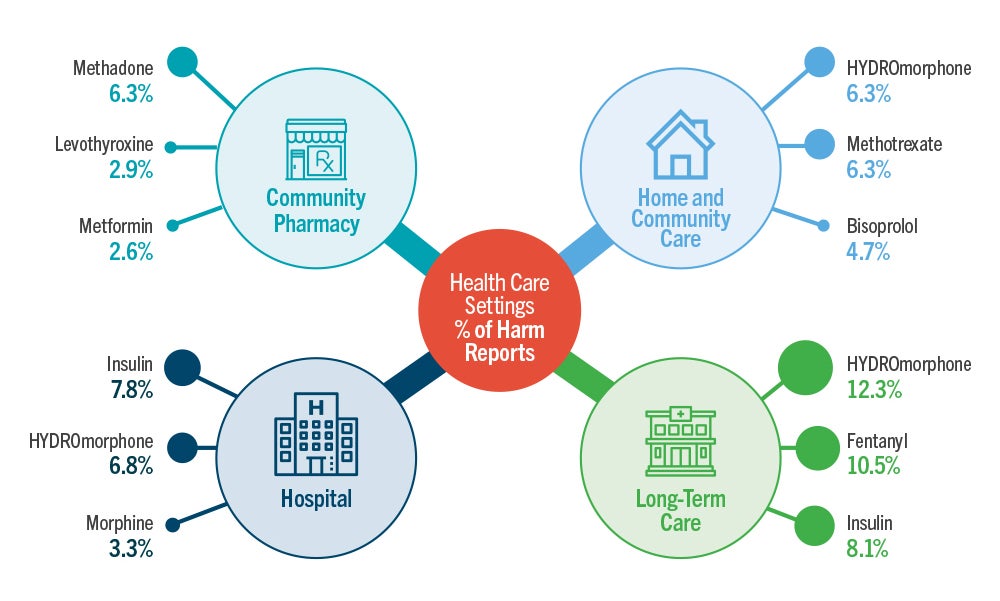
The top three medications cited in reports of harm incidents in each health-care setting are captured. Two medications appear in the top three in multiple settings: hydromorphone in all except community pharmacy and insulin in the two institutional care settings. Notably, these two medications were cited twice as often as any other medication in harm incidents from all health care settings combined (Table 1A).
ISMP has a vision of Zero Preventable Harm from Medications. To help attain this goal, the organization is a partner in the Canadian Medication Incident Reporting and Prevention System (CMIRPS). Other partners include Health Canada, the Canadian Institute for Health Information (CIHI), and the Canadian Patient Safety Institute (CPSI).
They compiled data from all health-care settings for the five-year period to identify the medications most frequently cited in reports of harm incidents with outcomes of any degree of severity (Table 1A) and those reported to have resulted in severe harm or death (Table 1B).
All five of the medications most frequently reported in severe harm or death incidents across all care settings over the past five years are defined as high-alert medications. For example, methotrexate for non-oncologic indications is considered to be a high-alert medication in the community and ambulatory setting. Contributing factors associated with severe harm related to methotrexate were dosage errors where doses prescribed for weekly administration were taken daily, unrecognized drug interactions, and inappropriately high doses prescribed for patients who were experiencing renal failure. Patients, families, and health-care providers may not appreciate the increased risks associated with this medication, nor be aware of the best practices recommended to prevent associated harm and death.
Although awareness of high-alert medications and the need for related safeguards increased over time, findings from the analyses serve as an opportune reminder that these and other medications continue to cause patient harm. There are known effective strategies for reducing the risk for error and harm for high-alert agents. Safety strategies and risk reduction measures, including optimization of technology, process improvement, patient/caregiver/health care provider education, and enhanced patient monitoring systems must be implemented.
The analyses highlight findings that merit further investigation:
- Hydromorphone was among the top three drugs reported in harm incidents in three of the four health care settings. Insulin was also frequently reported in harm incidents across multiple health care settings.
- In the community pharmacy setting, methadone was associated with the highest number of harm incidents. Insufficient patient identification processes, the need for individualized dosing regimens, and complex preparation steps were all contributing factors identified.
- Two medications reported as causing harm (acetaminophen and furosemide in Table 1A) are not identified high-alert drugs. Some reports that included acetaminophen involved a combination product containing this drug and an opioid (e.g., codeine, oxycodone).
- The appearance of levothyroxine in the list of incidents from community pharmacies may be influenced by the frequency of prescribing and dispensing this drug. Nonetheless, its presence in harm reports merits investigation into the root causes and contributing factors leading to patient harm.
ISMP Canada acknowledged that the reports submitted likely represent only a subset of the actual medication errors that are occurring. It is not possible to infer or project the probability of incidents based on voluntary reporting systems.
The value of reporting medication errors to CMIRPS for national analyses and shared learning is increasingly being recognized across health care settings. The findings from these analyses provide important insights into medications most frequently reported in association with preventable harm and death in Canada.
Reprinted with permission from ISMP Canada. ISMP Canada (www.ismp-canada.org) is an independent, national, not-for-profit organization with a mandate that includes analyzing medication errors and making recommendations to prevent harmful incidents.
Medications* most frequently reported in harm incidents with outcomes of any degree of severity, across all health care settings (Table 1A, top) and medications most frequently reported in severe harm or death incidents, across all health care settings (Table 1B, bottom), over a five-year period from Jan. 27, 2015 to Jan. 26, 2020
| Drug Name | Number of Incidents | % of Harm Reports |
|---|---|---|
| Insulin | 459 | 6.1% |
| HYDROmorphone | 447 | 5.9% |
| Morphine | 211 | 2.8% |
| Acetaminophen* | 199 | 2.6% |
| Methadone | 198 | 2.6% |
| Fentanyl | 172 | 2.3% |
| Heparin | 167 | 2.2% |
| Furosemide | 156 | 2.1% |
| Metoprolol | 152 | 2% |
| Warfarin | 137 | 1.8% |
*Any incident involving a combination product was counted toward the total for each of the active medicinal ingredients (e.g., an incident involving Percocet was counted with the totals for both oxycodone and acetaminophen).
| Drug Name | Number of Incidents | % of Harm Reports |
|---|---|---|
| HYDROmorphone | 27 | 11.1% |
| Morphine | 16 | 6.6% |
| Methotrexate | 13 | 5.3% |
| Methadone | 11 | 4.5% |
| Lorazepam | 7 | 2.9% |
Accessible summary
In terms of reports by degree of harm, 1.2% of incidents resulted in death, 2.1% of incidents were severe, 11.2% of incidents were moderate and 85.5% of incidents were mild.
Hospitals reported the most incidents (65.9%), followed by community pharmacy (26.5%), long-term care (6.7%), and home and community care (0.8%).












